Solution accelerator
Adverse Drug Event Detection
Pre-built code, sample data and step-by-step instructions ready to go in a Databricks notebook

Monitor drug safety with real-world data and NLP
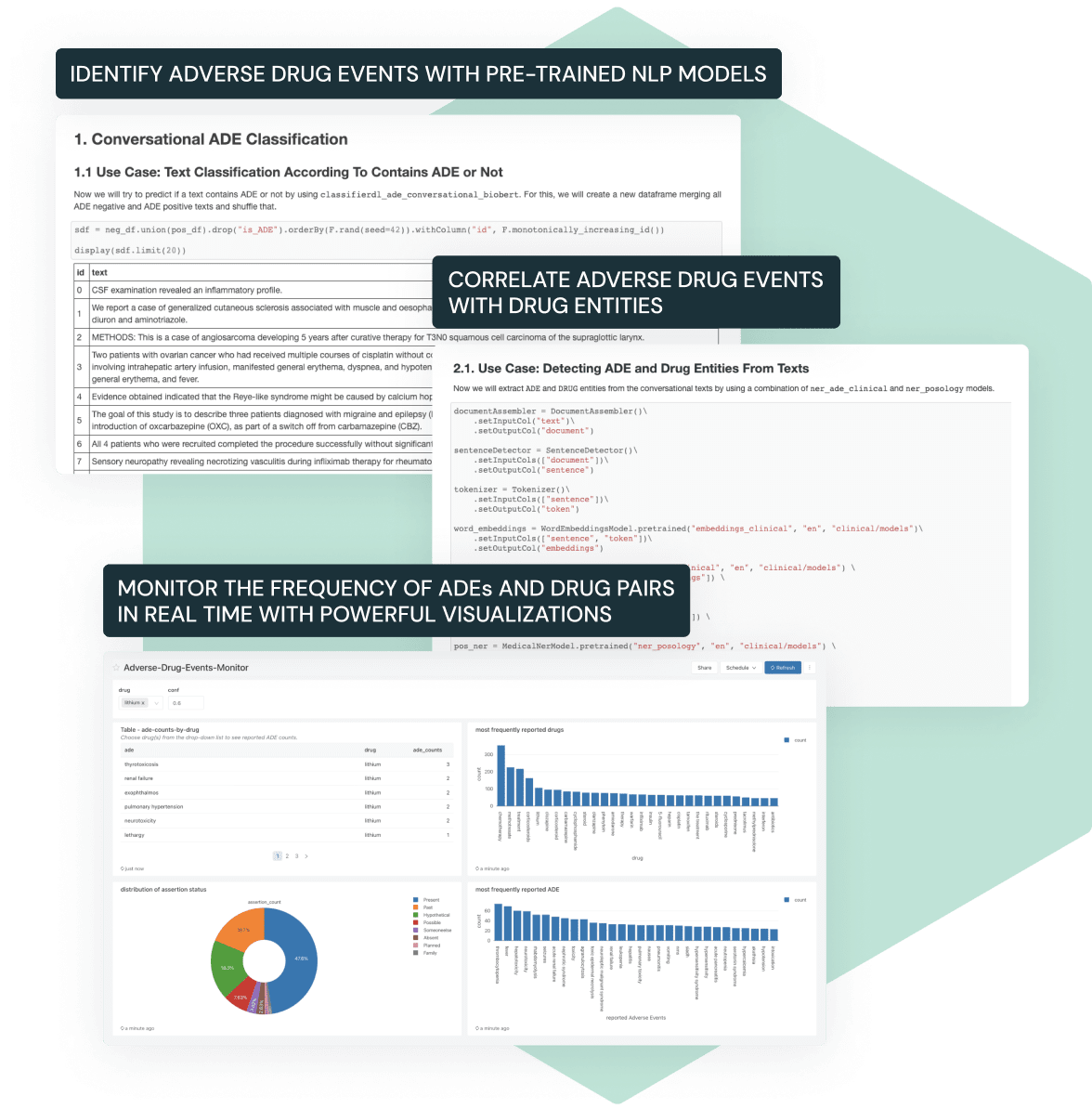
To ensure ongoing drug safety, pharma companies need to monitor and report adverse drug events post-market launch. This is challenging given that most adverse events are captured in unstructured text like emails, phone calls and social media posts. Our joint Solution Accelerator with John Snow Labs makes it easy to extract, process and analyze adverse drug events from real-world text data using natural language processing (NLP).
- Easily extract adverse events from large volumes of text data with NLP
- Automatically classify and correlate adverse events and drug entities
- Use built-in visualizations to assess the frequency of events
Resources
Blog

On-demand workshop

Blog

Deliver AI innovation faster with solution accelerators for popular industry use cases. See our full library of solutions
Ready to get started?